Because F is more electronegative than N, the bond moments (dipoles) are directed in the opposite sense to the lone-pair, so that the dipole moment of NF 3 is 0.234 Debye, compared with the value of 1.47 Debye of ammonia. 
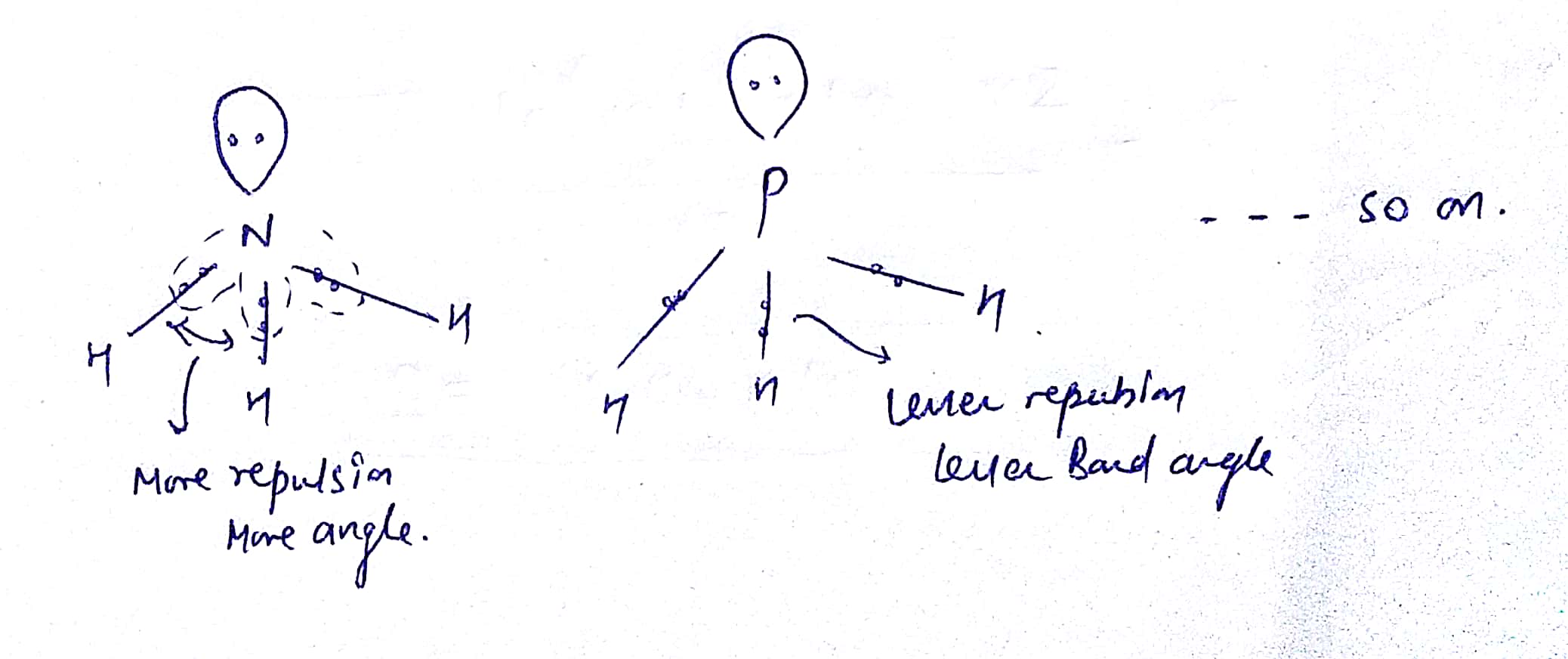
Why is that?Īgain, it’s the high electronegativity of fluorine that is responsible. Unlike ammonia, it does not act as a ligand to transition metals and form complexes. It is less toxic to inhale than nitrogen oxides (NO x) but does oxidise haemoglobin to methaemoglobin (which reduces oxygen carrying in the blood). It does not react with H 2 at 350☌, though the mixture explodes when sparked. NF 3 is only slightly soluble in water, it does not react with either water or dilute acid or alkali, nor with glass or mercury, for that matter. Up to 200 ☌, it is described as having similar reactivity to oxygen, but above that temperature it tends to dissociate appreciably into NF 2 and F radicals, turning it into a strong oxidising agent. The bond angle is reduced from 107° in ammonia to 101.9° in NF 3, because the very electronegative fluorines pull the electrons in the N-F bonds towards themselves, reducing interelectronic repulsions, so that the NF 3 ‘umbrella’ closes up.Īlthough NF 3 is quite stable at room temperature, its reactivity alters on heating. The molecule has a trigonal pyramidal structure, like ammonia. It is a colourless and odourless gas at room temperature, boiling at -129☌. NF 3 can also be made by reaction of ammonia with fluorine:Ĥ NH 3(g) 3 F 2(g) NF 3(g) 3 NH 4F(s) What’s it like? Otto Ruff (1871-1939, photo, right) was one of the great fluorine chemists of all time. 
The original discoverers (Ruff, Fischer and Luft, 1928) made it by electrolysis of a molten mixture of hydrogen fluoride and ammonium fluoride. Another factor is that fluorine is smaller than the other halogens there are likely to be large halogen-halogen repulsions in the other NX 3 molecules due to the difficulty in fitting three of them round a small nitrogen atom. The main reason for the favourable value for NF 3 is that the F-F bond is exceptionally weak compared to the other halogens (traditionally ascribed to non-bonding electronic repulsions in the F 2 molecule). In contrast, the enthalpy of formation of NCl 3 is 232 kJ mol -1 (259 using these figures) using E(N-Cl) = 192 kJ mol -1 and E(Cl-Cl) = 242 kJ mol -1. Unlike the other NX 3 molecules, NF 3 is an exothermic compound, its enthalpy of formation is -123 kJ mol -1. No, it is the only NX 3 molecule not to be explosive. 
I suppose this is explosive, too, like NCl 3 and NI 3? NF 3 is used as an etch gas to pattern the silicon chips. Nitrogen trifluoride The etching gas that's recently been foundĪ cleanroom used for fabrication of microelectronics. Going back to your question, we are supposed to compare $\ce$ also.Nitrogen trifluoride - Molecule of the Month - October 2019 (JSMol version) You can think of how s orbitals are spherical in shape, and a large s character would lead to more "spherical" hybrid orbitals, and that would lead to larger bond angles. What has all of this got to do with bond angles? Well, more s character leads to larger bond angles. Thus, Bent's rule can just as easily be formulated as follows: p character concentrates in orbitals directed towards electronegative substituents (the central atom doesn't have to waste its low energy s orbitals, the orbitals on the electronegative atom, can (possibly) take care of it. Which is why molecules like to use them (s orbitals) in bonds where there is "more" electron density to stabilise. Since s orbitals are lower in energy than p orbitals, they are better at stabilising electrons. Before that, I'll note that we concern ourselves with the hybridisation of the orbitals at the central atom. What follows below is a crude explanation. This can be argued on the basis of Bent's rule concisely statedĪtomic s character concentrates in orbitals directed toward electropositive substituents 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
